ISO/IEC 17025:2017 Lead Auditor Training
PAS301 IACET CEUs:4.0 Type: E-Learning

Description
This ISO/IEC17025 lead auditor training course, which comprises lectures, handouts, videos, audit checklist, and exams, is designed to provide training and certification to become ISO/IEC 17025 lead auditor for Laboratory Management System. This ISO/IEC17025 lead auditor training is an online course; you will learn everything you need to know as the ISO/IEC17025 lead auditor. You can receive this training from your office or home or anywhere through your computer and internet at your convenient time with multiple login options. This ISO/IEC17025 lead auditor training is immensely useful for busy laboratory technical team members, as they can attend this e-learning course from their home or office at their own convenient time.
With this ISO/IEC17025 lead auditor training course, the participants will gain knowledge and understanding of various activities involved in the audit. They will know Audit Techniques, Audit Process, and Audit Requirements as per iso 19011:2018. This course acknowledges them with how to conduct an opening meeting, perform an external audit as well as to conduct a closing meeting in any organization. Our ISO 17025 lead auditor training course is the first e-learning course in the world to include video of real-life examples on laboratory management system audit. This comprehensive course will help the laboratory employees as well as certifying body auditors to qualify as 'Lead Auditor' for Laboratory Management System based on ISO/IEC17025:2017 and get lead auditor certification.
Upon successful completion of this ISO/IEC17025 lead auditor training course, the participants will get a certificate for trained ISO/IEC17025:2017 Lead Auditor.
Topics Covered Under ISO/IEC17025 Lead Auditor Training
This ISO 17025 lead auditor training online course comprises four sections, as given below:
- Lectures:
There is a total of nine lecture sessions, which are given in this course as a ppt presentation with explanatory audio and visuals and graphics to understand the subject. The topics discussed in lecture sessions are listed below:- Session 1: Awareness of ISO/IEC 17025:2017
- Session 2: ISO 17025:2017 Lab Accreditation Process
- Session 3: Requirements of ISO/IEC 17025:2017 Laboratory Management System
- Session 4: Documented Information for ISO/IEC 17025:2017 Implementation
- Session 5: Risk-based approach ISO/IEC 17025:2017
- Session 6: ISO/IEC17025:2017Audit Process
- Session 7: Audit Terms and Definitions & Roles and Responsibilities.
- Session 8: Performing an Audit based on ISO/IEC 17025:2017
- Session 9: Nonconformity and Corrective Action.
- Handouts
For all the above nine sessions, the handouts are given in more than a total of 100 pages in pdf format. The participants of this ISO/IEC17025 lead auditor training can download and save the handouts in their computer and print them or read them offline to get detailed knowledge of all the nine topics mentioned above.- Manaul - In Session 8, a practical example of real-life laboratory management system manual is given for performing document review and understanding adequacy audit findings. Such real-life case study of a ISO/IEC 17025 manual will help participants to gain better knowledge and skill of auditing.
- Audit Checklist - A complete set of more than 400 audit questions, ISO/IEC17025:2017 requirement-wise as well as department-wise, are given as a ready tool to perform the lead audit. The ISO/IEC17025:2017 audit checklist also helps participants to prepare for the final audit. The participants can download it for future reference and use it during the audit process of the organization.
- Videos
This part of the training provides online training video tutorials of an overview of the standard, documentation, audit process, etc, all-important sessions of this course are covered and delivered by an experienced tutor. - Exams
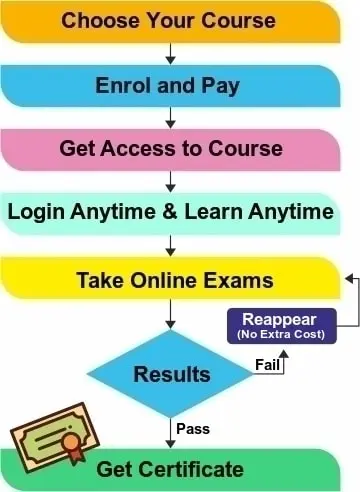
In this course, the participant has to clear a total of nine session exams and one final exam as given in a separate section of the course. After successfully passing the final exam, his/her auto-generated ISO/IEC17025 Lead Auditor Certificate will be issued and can be downloaded and printed from the dashboard.
Course Objectives
After completion of this ISO/IEC17025 auditor training e-learning course, you will be able to:
- Familiarize yourself with ISO/IEC17025:2017 Laboratory Accreditation principles.
- Understand the Plan-Do-Check-Act cycle of standard requirements.
- Familiarize yourself with the high-level structure and the framework of the laboratory management system.
- Understand the ISO/IEC17025:2017 requirements clauses and sub-clauses.
- Understand organizational issues and context of the organization.
- Know about leadership, planning, and support clauses.
- Know about technical requirements in testing and calibration laboratories.
- Know about the operation and performance evaluation clauses.
- Understand how improvement can be achieved in the laboratory and learn to audit with real-time audit examples.
- Understand, maintain and retain documented information list.
- Get the knowledge of external auditing and the use of audit checklist.
- Understand the processes involved in auditing, including opening meeting and closing meeting and requirements of ISO 19011 for auditing of management system standards.
- Know about the types of auditing and questioning techniques.
- Understand how to prepare and maintain internal audit records.
- Understand the new concepts of risk management and risk evaluation techniques.
Who should attend this course?
Any professionals working with laboratories or those with ISO/IEC17025:2017 accreditation bodies should attend this ISO/IEC 17025 auditor training. This online training can be beneficial for the following:
- Personnel of test and calibration laboratories.Quality managers of laboratories
- Laboratory managers
- Director/ Top management of laboratories and their representatives
- ISO/IEC 17025 consultants, compliance executives, experts
- Anyone who intend to perform audits conforming to the ISO/IEC 17025 standard.
This course is developed and brought to you by Punyam Academy, an Exemplar Global recognized training provider company. Now we are offering this ISO/IEC17025:2017 Lead Auditor training for working professionals, laboratory personnel, QA Assessors, Quality Managers, and other individuals for enhancing their career to new heights by becoming ISO/IEC17025:2017 Lead Auditor. Our e-learning courses help them to succeed in today's competitive environment, to renew licenses, and to update, strengthen and add quality to their existing knowledge and skills. Our courses are also useful for those who want to get a certification or start a new profession as a lead auditor.
This course has been created by a team of our consultants having experience of more than 20 years in ISO/IEC17025 management system consultancy in more than 35 countries and having experience of successful ISO/IEC 17025 accreditation for laboratories across the world, as well as having vast experience of designing and conducting various e-learning courses and training programs in convenient and cost-effective ways.
If you are a working professional, a student, an entrepreneur, or an individual looking for ISO/IEC17025 lead auditor certification online, Enrol Now!
Prerequisites
Following are considered as an added advantage for participants of this ISO/IEC17025 Lead Auditor Training course:
- Knowledge of Quality management systems.
- Understand the Plan-Do-Check-Act (PDCA) cycle.
- Knowledge of the following laboratory management system principles and concepts:
- 2 years of experience in a laboratory
- Ability to understand instructions in simple English
- People having a Diploma or Bachelor's degree definitely have an edge over the less qualified participant.
AUTHORS & INSTRUCTORS
For details of the authors, trainers and instructors experience and background, please visit our trainers page. They have very rich experience on the subject.
STUDY MATERIALS
This e-Learning course is provided with study materials and you can read it after your log in or download (PDF format). Use the study materials to reinforce key points and to keep a reminder of what you already learned as well as you can save it in your computer for future reference. The access of this study material is removed after exam is cleared and on line certificate is prepared for the student.
EXAMINATION AND COURSE CERTIFICATE
All paid E-learning course includes the Course Certificate that is issued upon course completion and passing the session exams as well as a final exam given in the course with minimum 60% marks. The option to reappear in the exam is also given to student, if failed in any exam.
END OF COURSE INSTRUCTION
After completing the session and passing each session examination the student can appear for final exam. Once the final exam is cleared then it is considered a end of course. The training certificate is ready on our LMS and student can print it or save the training certificate. It is also available for verification by entering the name and certificate number.

- Duration40 Hours (05 Days)
- Price (USD) $445
- IACET CEUs:4.0
- DOWNLOAD COURSE DEMO
- TypeE-Learning
- LanguageEnglish
Search Course
Why Learn with us?

How Automated LMS Works?